Introduction about TopSENSI ® STD-12 qPCR Kit
Currently, Sexually transmitted diseases – STD also known as Sexually Transmitted Infections – STIs) are becoming increasingly common across all genders and ages. Among them, individuals of reproductive age are the highest-risk group. STDs are typically transmitted from person to person through sexual intercourse (vaginal, oral, or anal), from mother to child, blood, and other bodily fluids. The agents causing STIs include fungi, bacteria, viruses, and parasites. When infected, patients may exhibit changing symptoms or be asymptomatic. If not detected and treated promptly, the disease can progress and lead to consequences such as urinary, genital tract inflammation, infertility, and even cancer development.
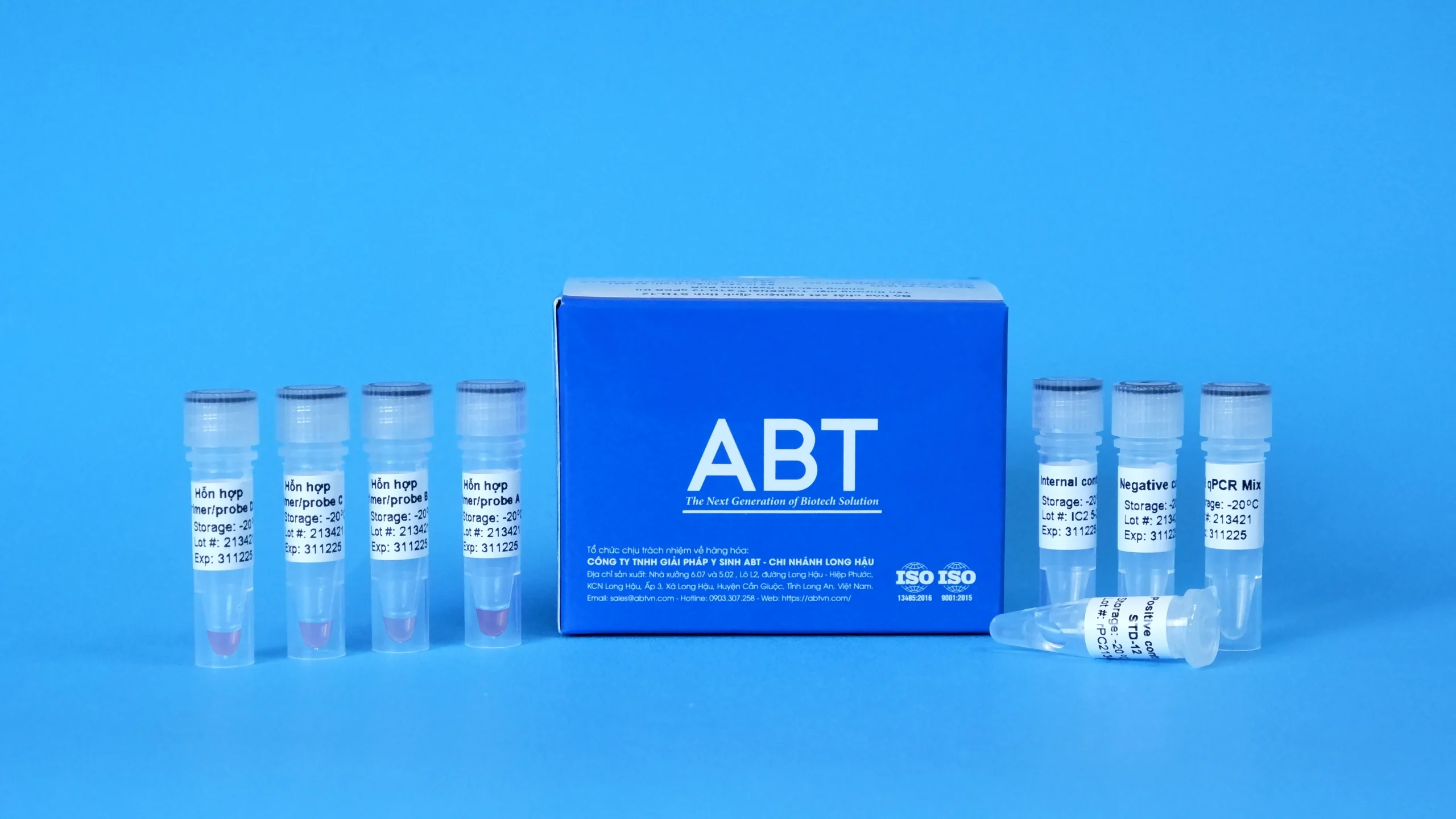
TopSENSI ® STD-12 qPCR Kit is a diagnostic assay used for the detection of 12 high-risk sexually transmitted pathogens, including Mycoplasma hominis; Mycoplasma genitalium; Trichomonas vaginalis; Ureaplasma parvum; Ureaplasma urealyticum; Gardnerella vaginalis; Human herpesvirus 1; Human herpesvirus 2; Treponema pallidum; Chlamydia trachomatis; Neisseria gonorrhoeae and Candida albicans The test is performed using Real-time PCR technology on DNA extracted from various human clinical specimens. The results obtained from this diagnostic kit are intended for clinical reference only and should not be used as the sole basis for diagnosis or exclusion of disease.
HIGHLIGHTS
- Rapid PCR execution (< 2 hours)
- High specificity and sensitivity
- Taqman probe technology
- Detection of multiple targets in a single qPCR reaction.
SPECIFICATIONS
| Targets | The kit detects 12 high-risk pathogens associated with sexually transmitted diseases, including: Mycoplasma hominis; Mycoplasma genitalium; Trichomonas vaginalis; Ureaplasma parvum; Ureaplasma urealyticum; Gardnerella vaginalis; Human herpesvirus I; Human herpesvirus 2; Treponema pallidum; Chlamydia trachomatis; Neisseria gonorrhoeae and Candida albicans |
| Input sample | Vaginal/urethral swab or cervical biopsy specimens. |
| Extraction sample volume | 200 µL |
| PCR sample volume | 10 µL |
| Fluorescent channels | FAM, HEX, TexasRed: for targets Cy5: for Internal control |
| Technology | TaqMan probe |
| PCR time | 1.5h |
| Analytical Specificity | Kit specifically detects 12 target sexually transmitted pathogens. No cross-reactivity with the host genetic material (human DNA) and some other microorganisms that may be present in the human body. |
| Storage | 12 months at -20oC |
| Kit components | STD-12 qPCR Mix, Primer/probe A Mixture, Primer/probe B Mixture, Primer/probe C Mixture, Primer/probe D Mixture, STD-12 Positive control, Negative control, Internal control (IC), PCR tube |
Analytical sensitivity

Recommended extraction kit
| Silica column extraction | TopPURE® Genomic DNA Extraction Kit |
| Silica column extraction | TopPURE® Fluid Viral Extraction Kit |
| Automatic magbead extraction | TopPURE® Maga Genomic DNA/RNA Extraction Kit |



Reviews
There are no reviews yet.