Information about cervical cancer and HPV Pap Smear Kit
Cervical cancer is a disease associated with abnormal proliferation of cells in the cervical epithelium (the organ between the uterus and the vagina), forming tumors in the uterus. Abnormal cervical cells exhibit characteristics such as multinucleation, increased pigmentation, and non-keratinization around the nucleus. The nucleus of abnormal cells is three times larger than that of normal cells. Changes in nuclear shape and cellular substance form the basis for cytological methods in UTCTC early screening.
Based on cell type, cervical cancer is divided into two main types: squamous cell carcinoma (SCC) and adenocarcinoma (AC). SCC occurs in the epithelial cells at the outer location of the cervix and is more common, while AC occurs in the endocrine glands of the cervix.
The main cause of cervical cancer is the Human papillomavirus (HPV), a virus that causes papillomas in humans and animals HPV (Human Papillomavirus) is transmitted sexually through unprotected sexual activity. According to the World Health Organization (WHO), cervical cancer is the fourth most common cancer among women worldwide. Globocan statistics report that 185 out of 204 countries worldwide have data on cervical cancer. Among them, Vietnam ranks 91st out of 185 for incidence rate and 50th out of 185 for mortality rate globally.
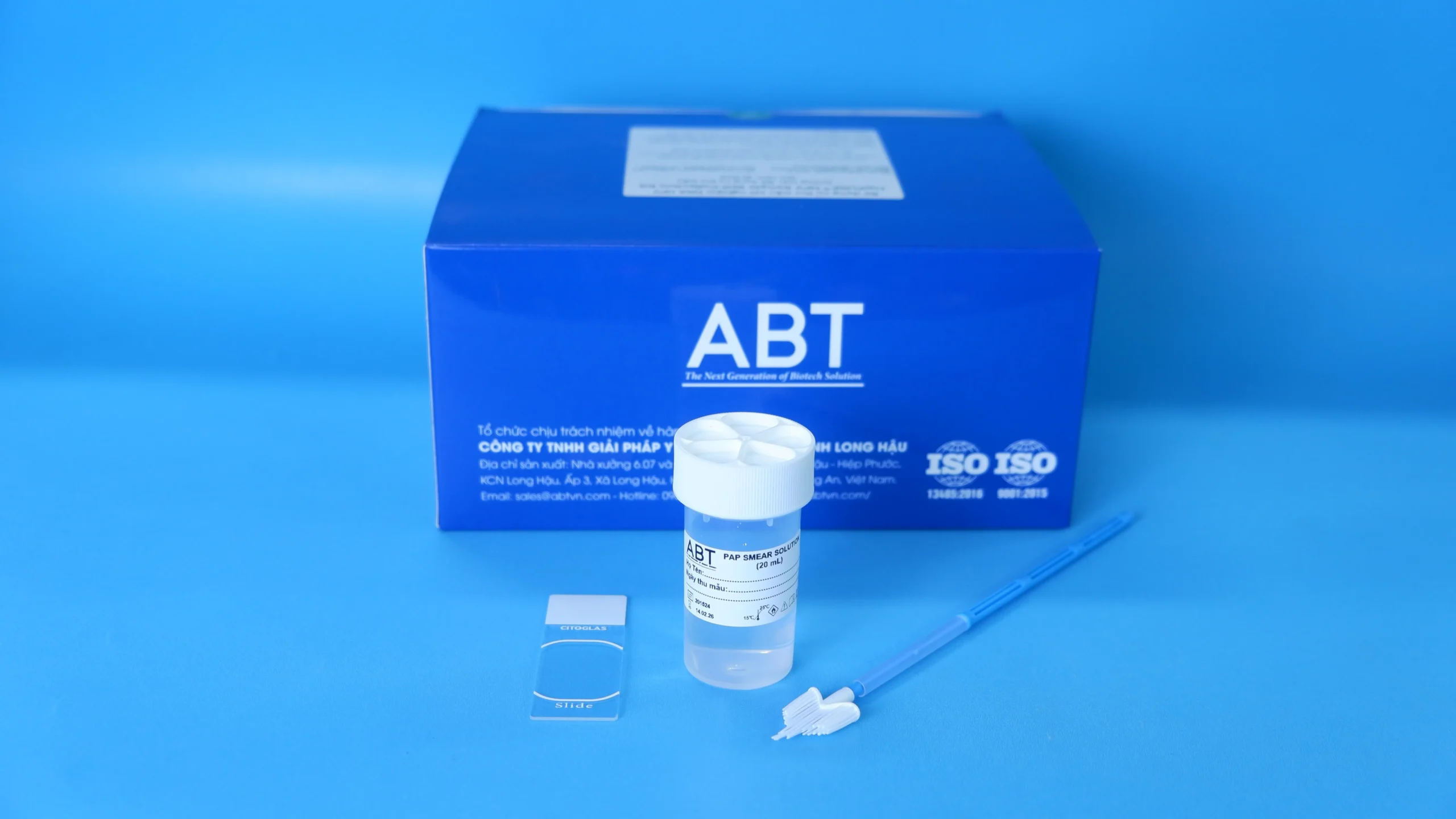
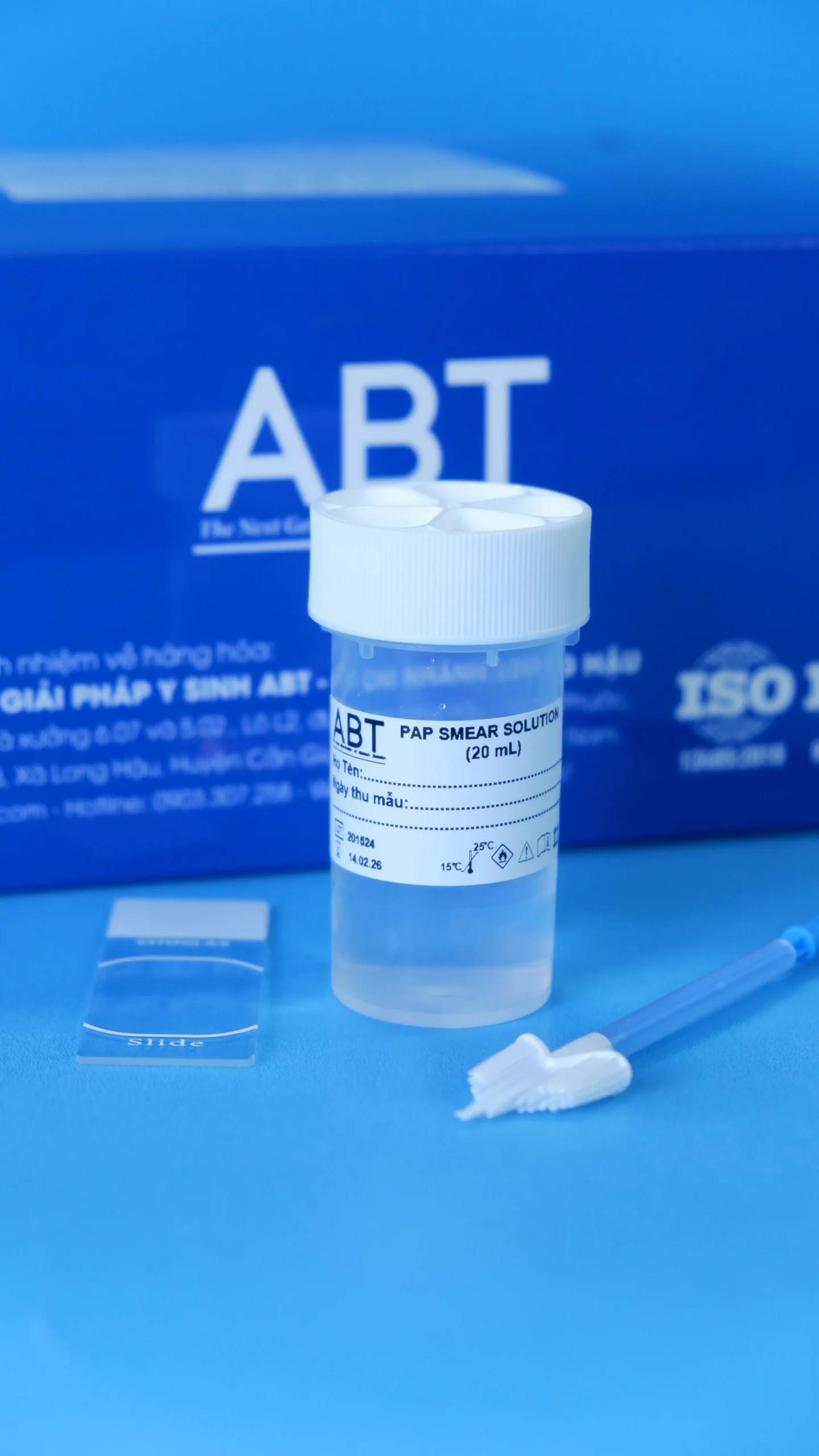
The TopPURE® HPV PAP SMEAR KIT (HI-017) is a set of tools designed for collecting and preserving vaginal cell samples for the purpose of cytological analysis in UTCTC screening (pap smear method). Additionally, the sample can be processed for the detection of HPV DNA. Kit HI-017 includes:
- The sampling stick NLD8303 consist of a handle part, a brush-type collecting part and remover tube part. The collecting part is made of PE (polyethylene) while other parts are made of PP (polypropylene). The sampling device is steriled by Ethylen oxide gas.
- Pap smear solution is transparent, colorless, solution pH 5.5 ± 0.2.
- Transparent glass microscope slide.
Manual instruction
- Collection sample procedure:
The collection of cervical smear samples will be carried out following the procedures outlined by the Ministry of Health.
- Step 1: Place the speculum in the vagina and adjust the position to ensure clear visibility of the cervix.
- Step 2: Insert the gynecological sampling brush into the cervical area, press gently, and rotate the brush clockwise 5 times (Figure 1).
- Step 3: Remove the brush from the vagina and gently rotate it in the Pap Smear Solution vial 5-10 times. Retain the brush head, collect the sample by detaching the head from the handle, and handle the brush appropriately.
- Step 4: Close the vial lid and record patient information on the label using a pencil.
Store and transport the sample at room temperature (25°C) for up to 1 month for cytology testing (Pap test) in cervical cancer screening.

– DNA-HPV extraction procedure:
Cervical swab samples preserved in Pap Smear Solution can also be used for qualitative HPV detection by Real-time PCR. DNA extraction and purification from samples preserved in Pap Smear Solution are performed as follows:
- Step 1: Aspirate 1 mL of the Pap Smear Solution-treated sample into a 1.5 mL tube. Centrifuge at 10,000 rpm for 3 minutes, collect the pellet, and discard the supernatant.
- Step 2: The pellet will be used for DNA extraction according to the kit protocol. TopPURE® Genomic DNA Extraction Kit (HI-112) - silica column extraction and TopPURE® Maga Genomic DNA/RNA Extraction Kit (HI-612) - magnetic beads extraction.
- Step 3: Qualify the DNA after extraction using TopSENSI® HPV qPCR Kit (SQH-103)
– Pap test: Use the microscope slide according to the device's instructions.
Note
- Shake Pap Smear Solution well before opening for use.
- Optimal sample collection time is before or after 2 days of the menstrual cycle.
- Not recommended for pregnant women.
Read more: HPV Genotype with RDB technique
For Real-time Kit: TopSENSI® HCV ONE-STEP RT-qPCR KIT CE (SQH-126)






Reviews
There are no reviews yet.