1. Information about Cervical Cancer and The HPV Sample-Self Collection Kit
Cervical cancer (CC) is a disease related to the abnormal proliferation of epithelial cells in the cervix (the organ connecting the uterus and the vagina), forming tumors in the uterus. Abnormal cervical cells exhibit the following characteristics: multinucleation, hyperchromasia, and vacuolization of the cytoplasm around the nucleus. The nuclei of abnormal cells are three times larger than those of normal cells. Changes in the shape of the nucleus and cytoplasm serve as the basis for cytological methods in early cervical cancer screening.
Based on cell type, cervical cancer is divided into two main types: squamous cell carcinoma (SCC) and adenocarcinoma (AC). SCC occurs in the epithelial cells at the outer location of the cervix and is more common, while AC occurs in the endocrine glands of the cervix.
The main cause of cervical cancer is the Human papillomavirus (HPV) is a virus that causes papillomas (warts) in humans and animals. HPV (Human Papillomavirus) is transmitted sexually through unprotected sexual activity. According to the World Health Organization (WHO), cervical cancer is the fourth most common cancer among women worldwide. Globocan statistics report that 185 out of 204 countries worldwide have data on cervical cancer. Among them, Vietnam ranks 91st out of 185 for incidence rate and 50th out of 185 for mortality rate globally.
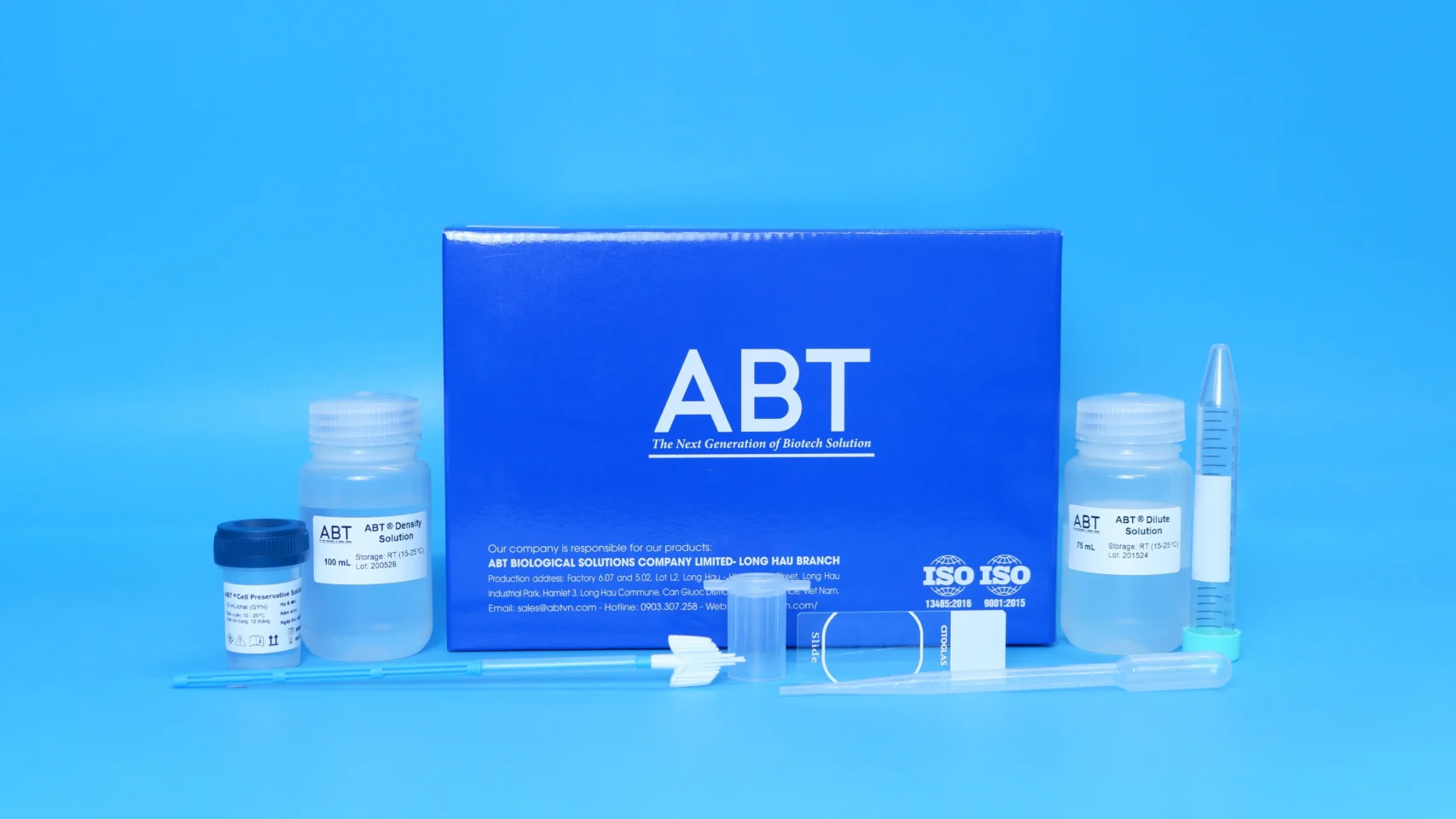
Bộ TopPURE ® HPV COLLECTION KIT is a kit designed for processing and collecting gynecological cells from cervical smear samples. The collected cervical sample is treated to remove mucus and blood in a buffered alcohol solution. The preserved cells remain morphologically stable (nucleus and cytoplasm) for at least one month at room temperature (15–25℃). In addition, the preserved cells can be used for qualitative HPV testing through HPV DNA detection. The HPV Sampling Kit includes:
- The sampling stick NLD8303 consist of a handle part, a brush-type collecting part and remover tube part. The collecting part is made of PE (polyethylene) while other parts are made of PP (polypropylene). The sampling device is steriled by Ethylen oxide gas.
- ABT® HPV Collection Medium: The collection medium is transparent and clearless, pH is 6.8 ± 0.2.
2. Manual intructions

3. Precaution
- Always wear gloves and a facemask during the procedure.
- Check the packaging and product carefully before use. If the product is damaged, do not use it.
- Each component in the kit is for single use only. Requirements of aseptic operations should be strictly followed. Discard immediately after use, do not reuse. Waste must be disposed in accordance with national environmental protection laws and regulations.
- The kit should be stored at a temperature of 15-25°C, relative humidity not more than 80%, good ventilation.
- The kit should avoid harsh condition such as high pressure, strong vibration, direct sunlight, high temperature, humidity, rain and corrosive gas or liquid during transportation.
- Carefully read the instructions for use, pay attention to the expiration date of the product before use. Do not use the product after the expiration date.
Read more: HPV Genotype with RDB technique
For Real-time Kit: TopSENSI ® HPV qPCR KIT (SQH-103)







Reviews
There are no reviews yet.