About TopSENSI ® STD-13 qPCR Kit
Sexually transmitted diseases (STDs), also referred to as sexually transmitted infections (STI), represent one of the most prevalent public‐health challenges worldwide. They rank among the top ten conditions prompting adults in developing countries to seek healthcare services. More than thirty distinct bacterial, viral, and parasitic agents can be transmitted via vaginal, anal, or oral intercourse, and certain STIs may also be vertically transmitted from mother to child during pregnancy, at birth, or through breastfeeding.
Group B Streptococcus (GBS) is a commensal bacterium commonly colonizing the vagina or rectum of 20–40% of women. Although GBS is not classified as an STD, individuals with a history of STI, those currently receiving antibiotic therapy, or those who tested positive for GBS in a previous pregnancy face a higher risk of GBS colonization in subsequent pregnancies compared to women without these factors. This association underscores the importance of comprehensive maternal health monitoring and medical history assessment during pregnancy, particularly regarding the interplay between GBS colonization and other reproductive‐tract infections such as STIs. Consequently, routine STD/STI screening is essential for early detection, appropriate treatment planning, and effective disease control.
TopSENSI ® STD-13 qPCR Kit s designed to detect thirteen high‐risk genital pathogens: Mycoplasma hominis; Mycoplasma genitalium; Trichomonas vaginalis; Streptococcus agalactiae; Ureaplasma parvum; Ureaplasma urealyticum; Gardnerella vaginalis; Human herpesvirus 1; Human herpesvirus 2; Treponema pallidum; Chlamydia trachomatis; Neisseria gonorrhoeae and Candida albicans The test is performed using Real-time PCR technology on DNA extracted from various human clinical specimens. The results obtained from this diagnostic kit are intended for clinical reference only and should not be used as the sole basis for diagnosis or exclusion of disease.

TopSENSI ® STD-13 qPCR Kit
HIGHLIGHTS
- Rapid PCR execution (< 2 hours)
- High specificity and sensitivity
- Taqman probe technology
- Detection of multiple targets in a single qPCR reaction.
SPECIFICATIONS
| Targets | Kit detects 14 high‐risk genital pathogens including: Mycoplasma hominis; Mycoplasma genitalium; Trichomonas vaginalis; Streptococcus agalactiae; Ureaplasma parvum; Ureaplasma urealyticum; Gardnerella vaginalis; Human herpesvirus I; Human herpesvirus 2; Treponema pallidum; Chlamydia trachomatis; Neisseria gonorrhoeae and Candida albicans |
| Input sample | Vaginal/urethral swab or cervical biopsy specimens. |
| PCR sample volume | 10 µL |
| Fluorescent channels | FAM, HEX, TexasRed và Cy5: for targets |
| Technology | TaqMan probe |
| PCR time | 1.5h |
| Analytical Specificity | Kit specifically detects 13 target sexually transmitted pathogens. No cross-reactivity with host genetic material (human DNA) or with other microbial agents potentially present in humans. |
| Storage | 12 months at -20oC |
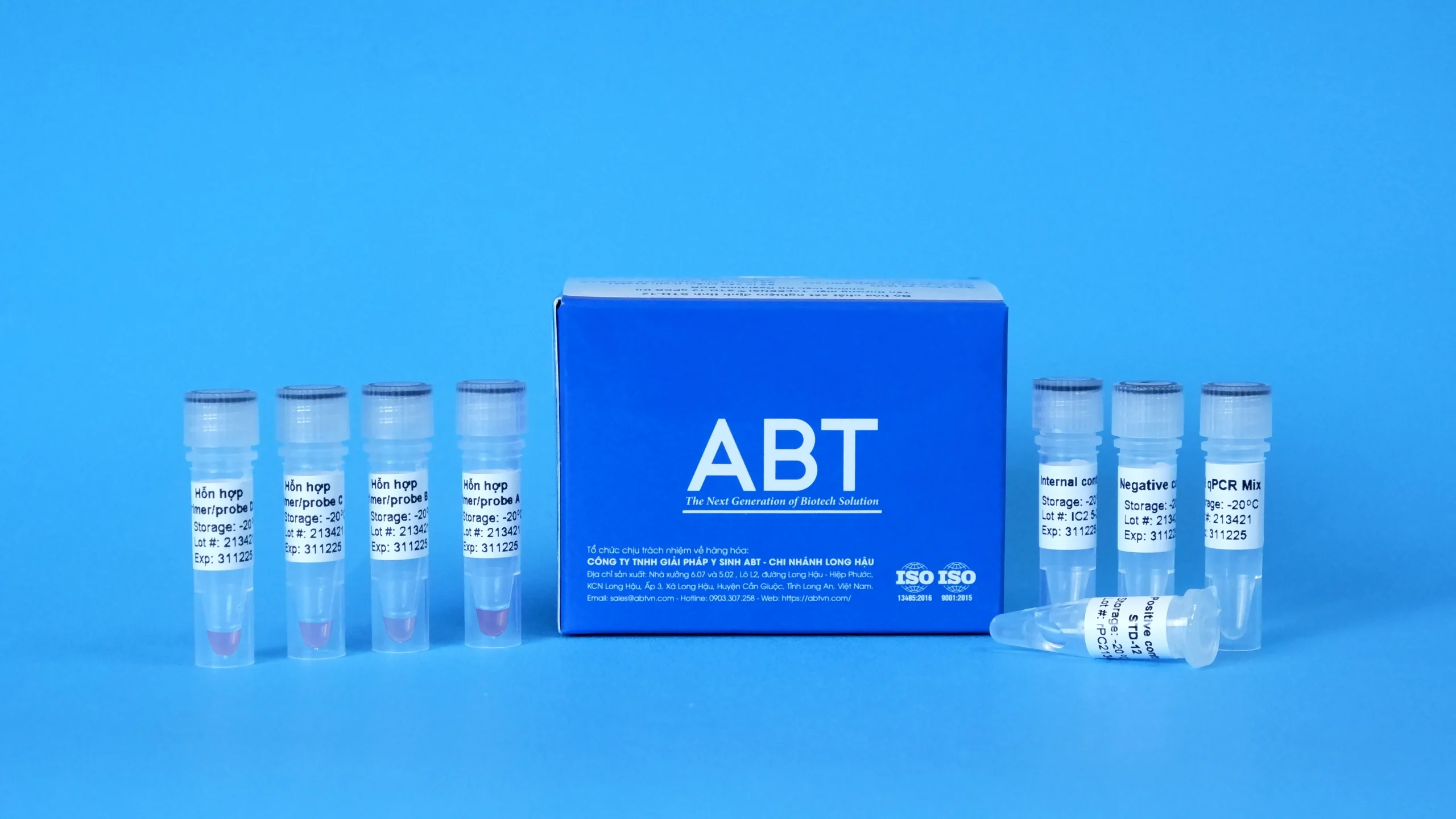
| Kit components | STD-13 qPCR Mix, Primer/probe A Mixture, Primer/probe B Mixture, Primer/probe C Mixture, Primer/probe D Mixture, Positive control, Negative control, Internal control, PCR tube |
Analytical sensitivity
| Group | Targets | Sensitivity (copies/µL) |
| A | Mycoplasma hominis | 1,50 |
| Mycoplasma genitalium | 1,00 | |
| Trichomonas vaginalis | 1,00 | |
| Streptococcus agalactiae | 1,00 | |
| B | Ureaplasma parvum | 1,50 |
| Ureaplasma urealyticum | 1,50 | |
| Gardnerella vaginalis | 1,00 | |
| C | Chlamydia trachomatis | 4,00 |
| Neisseria gonorrhoeae | 4,00 | |
| Candida albicans | 2,50 | |
| D | Human herpesvirus I | 1,00 |
| Human herpesvirus II | 3,50 | |
| Treponema pallidum | 2,00 |
Recommended extraction kit
| Silica column extraction | TopPURE® Genomic DNA Extraction Kit |
| TopPURE ® Fluid Viral Extraction Kit | |
| TopPURE® Serum Viral DNA/RNA Co-Extraction Kit | |
| Automatic magbead extraction | TopPURE® Maga Genomic DNA/RNA Extraction Kit |
| TopPURE® Maga Serum DNA/RNA Co-Extraction Kit |



Reviews
There are no reviews yet.