Information about cervical cancer and HPV Collection Kit
Cervical cancer (CC) is a disease related to the abnormal proliferation of epithelial cells in the cervix (the organ connecting the uterus and the vagina), forming tumors in the uterus. Abnormal cervical cells exhibit the following characteristics: multinucleation, hyperchromasia, and vacuolization of the cytoplasm around the nucleus. The nuclei of abnormal cells are three times larger than those of normal cells. Changes in the shape of the nucleus and cytoplasm serve as the basis for cytological methods in early cervical cancer screening.
Based on cell type, cervical cancer is divided into two main types: squamous cell carcinoma (SCC) and adenocarcinoma (AC). SCC occurs in the epithelial cells at the outer location of the cervix and is more common, while AC occurs in the endocrine glands of the cervix.
The main cause of cervical cancer is the Human papillomavirus (HPV) is a virus that causes papillomas (warts) in humans and animals. HPV (Human Papillomavirus) is transmitted sexually through unprotected sexual activity. According to the World Health Organization (WHO), cervical cancer is the fourth most common cancer among women worldwide. Globocan statistics report that 185 out of 204 countries worldwide have data on cervical cancer. Among them, Vietnam ranks 91st out of 185 for incidence rate and 50th out of 185 for mortality rate globally.
The TopPURE ® HPV COLLECTION KIT (HI-018) is a product optimally designed for the collection, preservation, and transportation of gynecological smear samples for use in qualitative real-time PCR (qPCR) testing Human papillomavirus (HPV). The HI-018 kit includes:
- Sample collection swab:
- Swab material: PA66 + PP + Stainless Steel 304
- Swab length: 195mm
- Sample transport medium:
- 2.5 mL of sample transport medium contained in a 5 mL tube, used for sample preservation.
- The solution is clear, colorless, with a pH of 6.8±0.5.
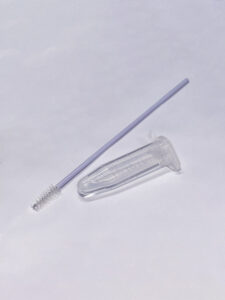
Kit components TopPURE ® HPV COLLECTION KIT (HI-018)
Manual instruction
- Step 1: Open the swab package partially and take out the swab. Avoid touching the swab head or allowing the swab to come into contact with any surface. If the swab head is touched, dropped, or compromised, use a new sampling kit.
- Step 2: After vaginal dilation, insert the swab head into the vagina until the brush bristles and cervix make contact. Hold the swab handle with the thumb and forefinger. Maintain gentle pressure, after rotating 5 turns in one direction, remove the sample swab without touching the skin.
- Step 3: Open the lid of the tube containing the transport medium. Be careful not to spill the solution in the tube. Place the swab into the container tube, and rotate 5 - 10 turns in one direction.
- Step 4: Remove the swab, tightly seal the tube cap, and clearly record the patient information.
Transport the specimen to the laboratory as soon as possible, not later than 7 days after collection.
Note
- Gloves and masks should be worn during the procedure.
- Avoid use during menstruation, pregnancy and lactation.
- It is not recommended for women with genital tract injuries or those who have recently undergone cervical surgery. Cervical inflammation should ideally be treated prior to sample collection, and sampling should occur post-recovery.
- Abstaining from sexual intercourse within 24 hours preceding sample collection is advisable.
- Vaginal douching or intravaginal medication should not be performed within 3 days prior to sample collection.
- Do not use if the packaging is damaged, the sampling tip is in contact with the tube or the sampling tip is separated from the tube, or the product has expired.
- In case of persistent bleeding or pain during the sampling procedure, immediate cessation is advised, and medical attention should be sought promptly.
- Waste must be treated according to legal documents and regulations on national environmental protection.
Read more: HPV Genotype with RDB technique
For Real-time Kit: TopSENSI® HPV qPCR kit








Reviews
There are no reviews yet.