About TopSPEC® Doxycycline MIC Test
Minimum Inhibitory Concentration (MIC) is the lowest concentration of an antibacterial substance that visibly inhibits the growth of microorganisms. MIC assists in selecting the most appropriate antibiotics, preventing antibiotic misuse, contributing to the reduction of bacterial antibiotic resistance, shortening treatment duration, saving costs, and enhancing treatment effectiveness.
TopSPEC® Doxycycline MIC Test is a diagnostic kit designed to determine the Minimum Inhibitory Concentration (MIC)—the lowest concentration of antibiotic capable of inhibiting bacterial growth.
Product description
Purpose of Use: To determine the Minimum Inhibitory Concentration (MIC)—the lowest concentration of antibiotic capable of inhibiting bacterial growth.
Components: Doxycycline antibiotic plates, 50X R staining reagent, Sterile water
Packaging: 16 tests
Product code: LAK-104
Storage Conditions: 2–8 °C
Expiration date: 12 months from the date of manufacture
Procedure
Step 1: Preparation
- Bacterial Sample: Culture bacterial colonies on non-selective agar plates for 18–24 hours. Prepare a bacterial suspension with a turbidity equivalent to McFarland 0.5.
- 50X R Staining Reagent:
- Spindown before manipulation
- Add the staining solution to sterile water at a ratio of 1:50
- Amount of solution used: 300µL/1 strip (1 test)
- Culture medium: Use CAMHB for easily cultured aerobic strains or LHB-CAMHB for fastidious or microaerophilic strains. (Note: Medium is not included in the kit.)
Step 2: Execution
- Prepare approximately 3 mL of medium per test
- Add 100 µL of medium to the NEG wells
- Dilute the McFarland 0.5 bacterial suspension at a ratio of 1:300 in the culture medium
- Inoculate 100 µL of the bacterial suspension into all wells except the NEG wells
- Incubate at 35–37 °C for 16 to 20 hours

Figure 1: Layout of Doxycycline Concentration Levels on a Single Plate (Concentration Unit: μg/mL)
*Note:
- NEG: Well containing only the medium
- POS: Well containing only the bacterial suspension
Results reading day:
- Add 25μL of staining solution R (diluted 1:50 in sterile water) to all wells
- Incubate the plate for 30-40 minutes at 35-37°C
- Read the MIC results.
Step 3: Results reading
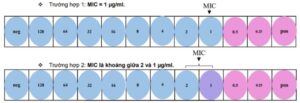
Figure 2: Representative outcomes following MIC testing.
- Negative control wells (NEG) with no color change (still showing the green color of the staining solution) ensure that the culture medium is not contaminated.
- Positive control wells (POS) exhibit a color change (pink or light pink), ensuring the stability of the staining solution and the normal growth of bacteria.
- Wells showing a color change from green to pink or no color change after adding the staining solution indicate the biochemical reactions of live bacteria, generating chemical reactions with the staining solution, resulting in a color change. This demonstrates the presence of viable bacteria.
- The well containing the lowest antibiotic concentration that shows no color change (retains a blue hue) after the addition of the staining reagent is considered the MIC value—the minimum inhibitory concentration capable of preventing bacterial growth. This lack of color change indicates insufficient viable bacteria to trigger the chemical reactions responsible for color transformation, thereby confirming the antibiotic’s inhibitory effect at that concentration.
- In cases where the color changes from green to purple (or not entirely pink), the MIC value is approximately between this antibiotic concentration and the higher neighboring concentration.
Read more:






Reviews
There are no reviews yet.