Information about Cervical Cancer and ABT ® Cervical Cytology Manual Kit
Cervical cancer is a disease associated with the abnormal proliferation of epithelial cells in the cervix (the organ connecting the uterus and the vagina), forming tumors in the uterus. Abnormal cervical cells exhibit the following characteristics: multinucleation, hyperchromasia, and cytoplasmic vacuolization around the nucleus. The nuclei of abnormal cells are three times larger than those of normal cells. Changes in nuclear and cytoplasmic morphology form the basis for cytological methods in early screening for cervical cancer. Based on the cell type, cervical cancer is classified into two main types: squamous cell carcinoma (SCC) and adenocarcinoma (AC). SCC occurs in the epithelial cells at the outer cervix and is more common, while AC occurs in the glandular cells of the cervix.
The main cause of cervical cancer is the Human papillomavirus (HPV), a virus that causes papillomas in humans and animals. HPV spreads through genital contact via unsafe sexual activities. According to the World Health Organization (WHO), cervical cancer is the fourth most common cancer among women worldwide. In 2020, there were approximately 604,000 new cases and 342,000 deaths from cervical cancer. About 90% of new cases and deaths occurred in low- and middle-income countries. According to Globocan statistics, 185 out of 204 countries worldwide report on the situation of cervical cancer. Vietnam ranks 91st out of 185 in terms of new incidence rate and 50th out of 185 in terms of mortality rate globally. This shows a significant increase compared to 2018, where the new incidence rate was 99th out of 185, and the mortality rate was 56th out of 185.
ABT® Cervical Cytology Manual Kit is a tool for collecting, processing, and spreading gynecological cells using centrifugation combined with natural sedimentation. Cervical smear samples are collected at the cervix, processed to remove mucus and blood from the sample matrix, and stabilized in an alcohol-based buffer solution for up to one month at a temperature range of 15–25°C, serving cytological testing purposes (liquid-based Pap test) for cervical cancer screening. Additionally, smear samples collected and preserved using the kit can be processed for HPV-DNA testing.
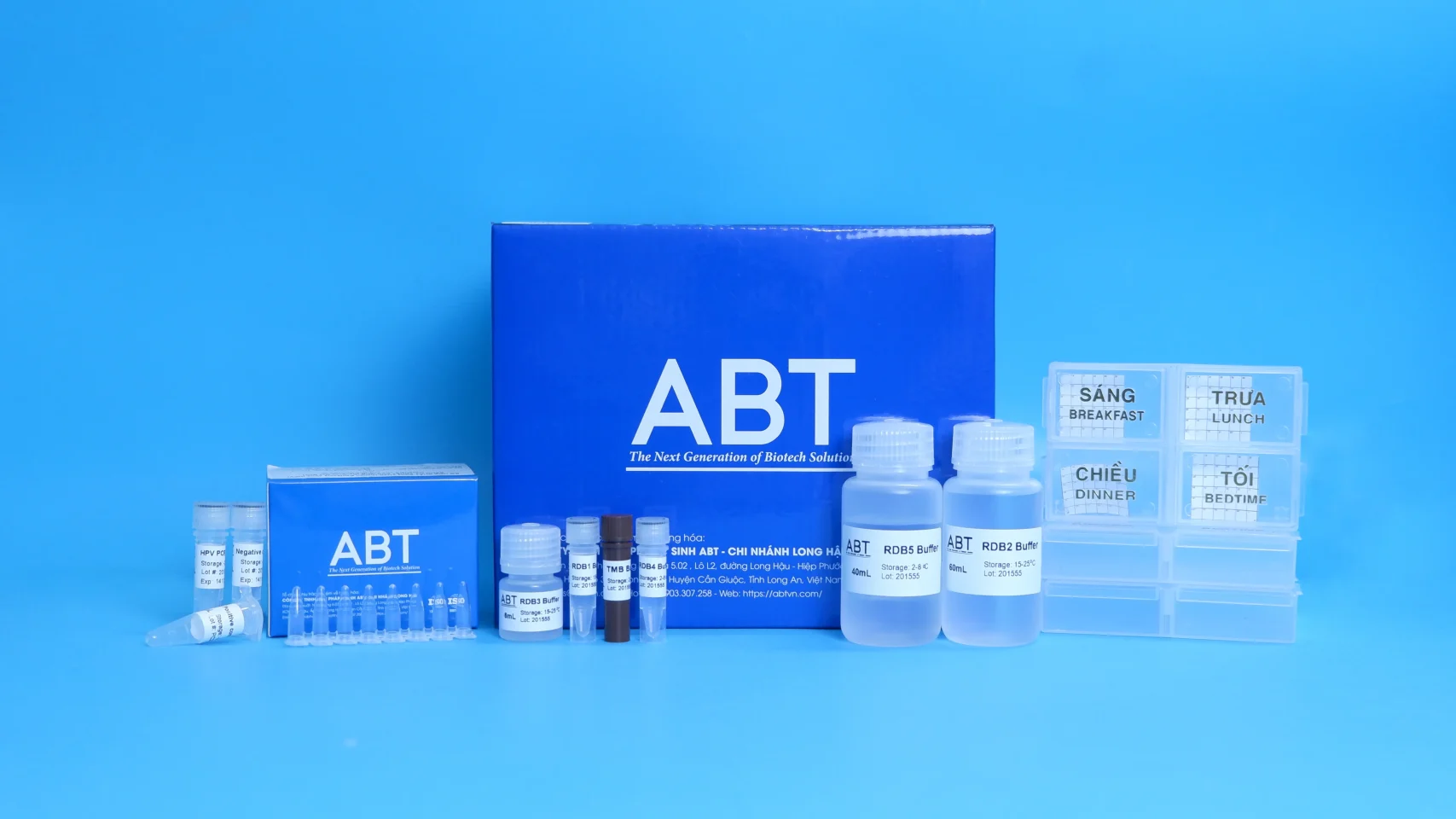
The ABT ® Cervical Cytology Manual Kit (DD-102) including:
- Sample collection swab:
- Material: PE + PP
- Sample collection head diameter: 19.5 ± 2 mm
- Total length: 202±10 mm
– ABT® Cell Preservative Solution:
- A homogeneous, transparent, colorless liquid, free from impurities and sediment. It has the characteristic odor of the product without any unusual smells. pH 5.5 ± 0.2.
- Sample storage duration: Within one month from the collection date.
- Cell sedimentation chamber
- Material: PE + PP
- Dimensions: 35 mm × 20 mm × 15 mm (Height × Body Diameter × Base Diameter)
- Microscope slide:
- Material: Glass
- Dimensions: 75 mm × 25 mm × 1 mm (Length × Width × Thickness)
– ABT ® Density Solution:
- A homogeneous, transparent, colorless liquid with slight viscosity, free from impurities and sediment. It has the characteristic odor of the product without any unusual smells.
- Processing capability: Processes and removes mucus, blood, and inflammatory cells in gynecological smear samples (GYN) and collects target epithelial cells.
– ABT ® Dilute Solution:
- A homogeneous, transparent, colorless liquid with slight viscosity, free from impurities and sediment. It has the characteristic odor of the product without any unusual smells.
- Processing capability: Separates and evenly distributes cells on the slide during smear preparation.
Manual instruction ABT ® Cervical Cytology Manual Kit
1. Sample preparation:
Cervical smear samples will be collected according to the Ministry of Health's procedure (Appendix 1, Decision 3877/QD-BYT 2019):
- Step 1: Insert a speculum into the vagina and adjust its position to ensure a clear view of the cervix.
- Step 2: Insert the sampling brush into the cervix, press gently, and rotate the brush clockwise 5 times (Figure 1).
- Step 3: Remove the sampling brush from the vagina and place it into the ABT® Cell Preservative Solution vial, gently rotating it 5 to 10 times. The head of the sampling brush is retained by detaching it from the handle, and the handle is disposed of properly.
- Step 4: Close the vial and record the patient's information on the label with a pencil.
Store and transport the sample at room temperature (15-25℃) for up to 1 month for cytological examination (liquid-based Pap test) in cervical cancer screening. Additionally, samples collected and preserved using the kit can be processed for HPV-DNA testing.
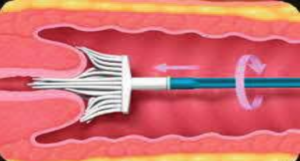
Figure 1 Position for placing the cervical smear sampling brush.
2. Processing and smearing slides
- Record patient information on the frosted edge of the electrostatic glass slide using a pencil and on the cell sedimentation chamber filter cup and plastic pipette using a marker.
- Proceed with sample processing and smear preparation using a Falcon centrifuge according to the following steps:
- Step 1:Vortex the sample thoroughly for 30 seconds and transfer 7 mL of the sample solution into a 15 mL Falcon tube.
- Step 2: Use a plastic pipette to aspirate 3 mL of ABT® Density Solution and carefully dispense it to the bottom of the tube, allowing it to gently settle so that the sample solution and ABT® Density Solution form two distinct layers.
- Step 3: Centrifuge at 4000 rpm for 10 minutes. Use a plastic pipette to remove approximately 7 mL of the supernatant, avoiding aspiration of the precipitate in the tube.
- Step 4: Centrifuge again at 4000 rpm for 5 minutes to collect the cell pellet. Use a plastic pipette to remove the remaining supernatant and retrieve the precipitate.
- Step 5: Add approximately 1–5 mL of ABT® Dilute Solution (maintaining a sample/ABT® Dilute Solution ratio of 1:20) to dilute the sample.
- Step 6: Assemble the glass slide and sedimentation chamber on the plastic tray. Pipette 500 µL of the diluted sample directly onto the sedimentation chamber and let it sit for 5 minutes.
- Step 7: Remove the supernatant and retrieve the glass slide containing the cells.
- After smearing, fix the glass slide in 96% ethanol for 15 minutes and stain with PAP according to the procedure of the ABT ® Papanicolaou Staining Kit (DD-083; DD-084).
- Observe and analyze the results under a microscope.
Note:
- Always wear gloves and a mask during the procedure.
– Shake the ABT® Cell Preservative Solution well before opening the cap for use.
– The appropriate sample collection time is within 2 days before or after menstruation.
– Contraindicated in pregnant women.
– Waste must be disposed of according to national environmental protection laws and regulations.
3. Recommendations:
Compatible with centrifuge models designed for 15 mL Falcon tubes.
Read more: TopPURE® HPV Pap Smear Kit (HI-017)






Reviews
There are no reviews yet.