Introduction TopSENSI ® HP Clotest
Helicobacter pylori a common stomach pathogen that causes gastritis, gastric ulcers, gastric adenocarcinoma, and low-grade gastric lymphoma. The infection can be asymptomatic or cause indigestion of varying degrees.
HP infection is one of the most common chronic bacterial infections in humans. It is estimated that over 50% of the population has been infected with HP, primarily in developing countries where infection rates are very high, ranging from 50-90% in individuals over the age of 20, and most children are infected between the ages of 2-8. Vietnam also has a high HP infection rate, around 70% in adults. In developed countries, the age of infection is typically over 50, affecting more than 50% of the population. This rate increases by 10% each year.
The primary transmission route is via the oral-fecal (fecal-oral) or direct (oral-oral) route through saliva. In areas with poor sanitation, contaminated water and food are the primary initial sources of transmission.
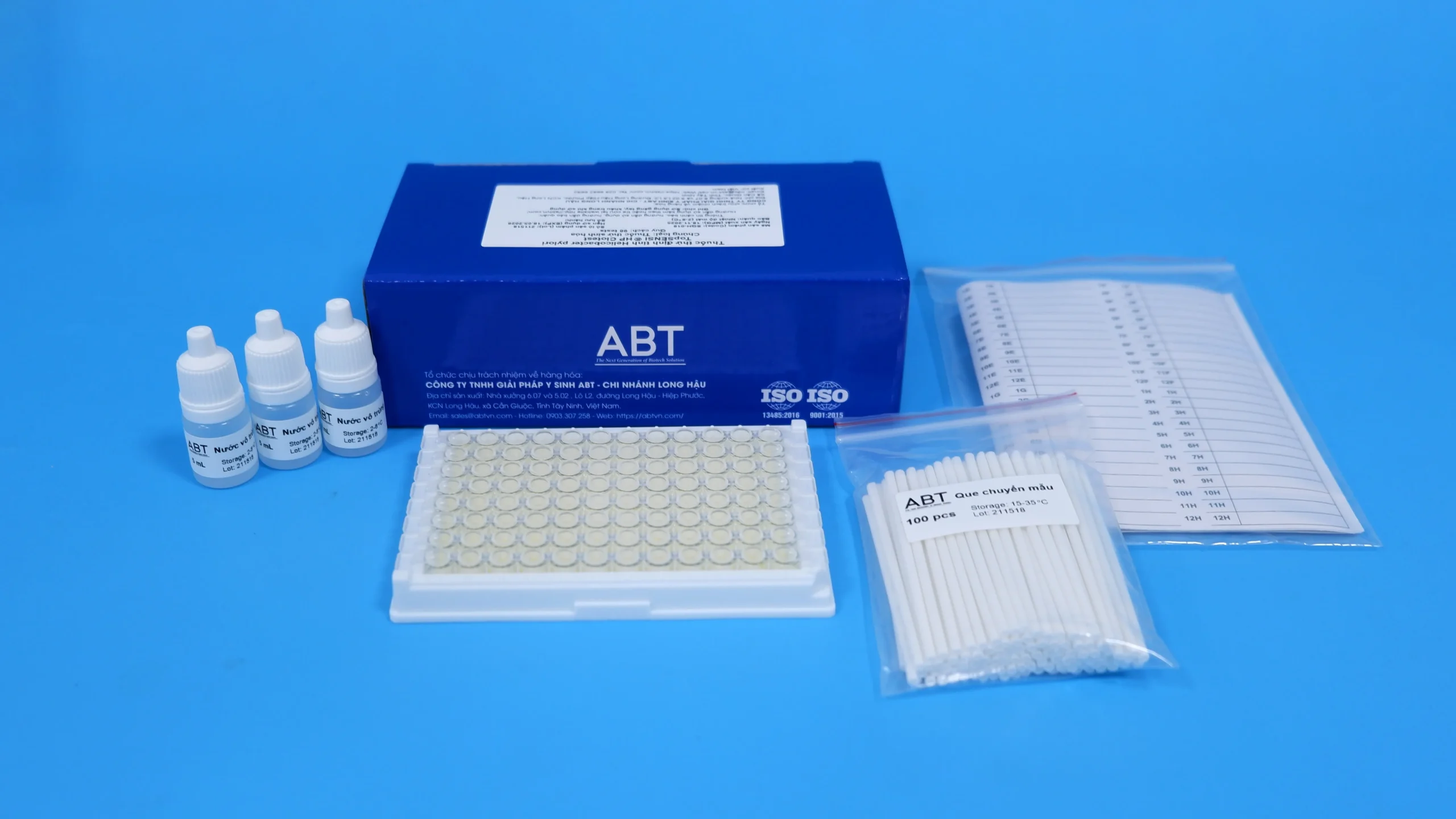
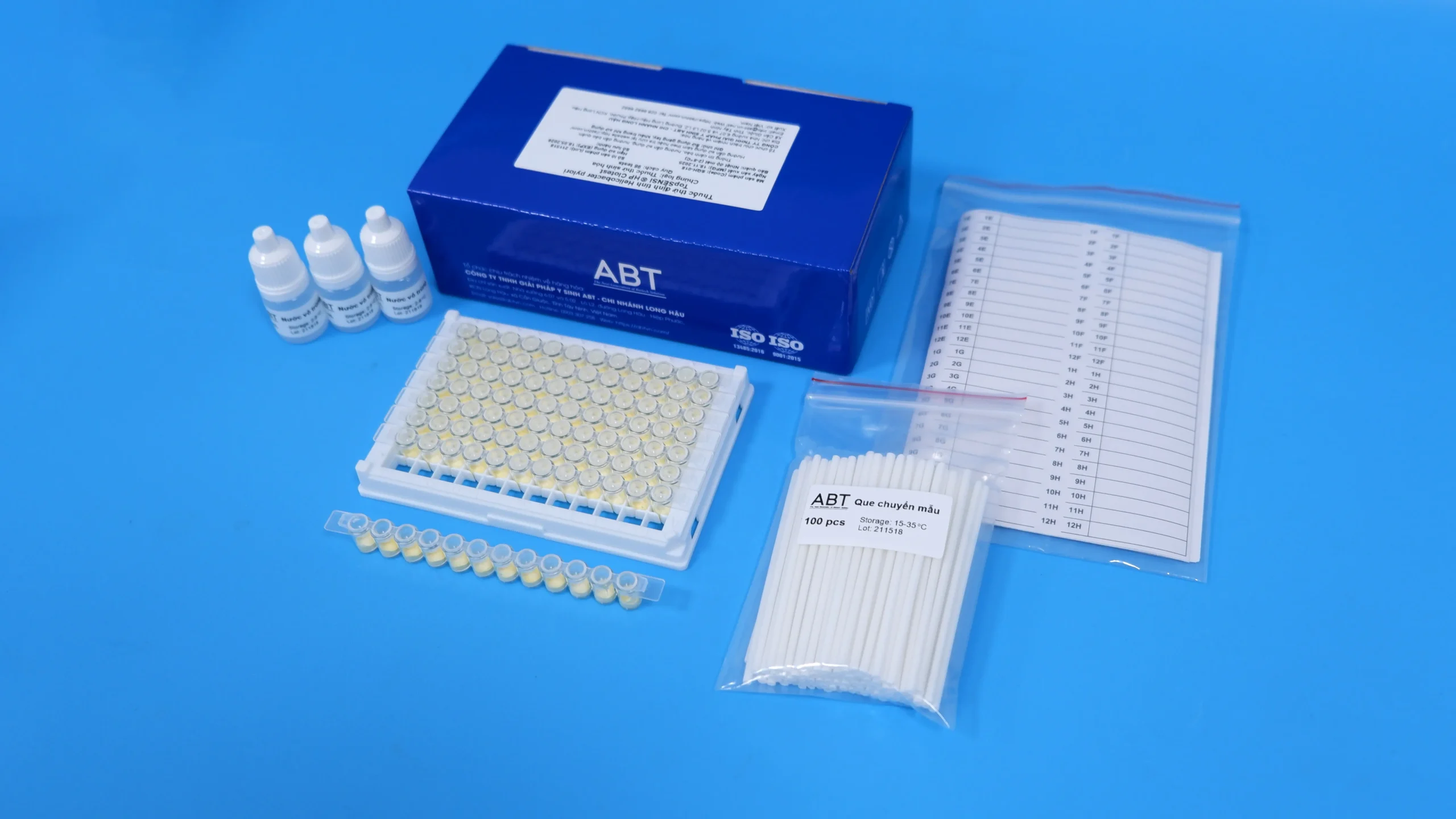
TopSENSI ® HP Clotest is a biochemical reagent designed for the detection of Helicobacter pylori from gastric biopsy specimens or culture media. The reagent is supplied in a solid medium. Upon reconstitution, the test undergoes a color change from yellow to pink within 30 minutes in the presence of H. pylori. The assay determines urease activity by immersing gastric biopsy specimens or culture media into the reagent. For testing bacterial cultures, 50 µL of the bacterial suspension is added to the reagent.
Highlights
- High stability:
- The product is provided in lyophilized form, which ensures greater stability compared to liquid or gel formulations, even when stored at room temperature.
- The reagent maintains consistent coloration, facilitating large-scale storage under cool conditions.
– Superior accuracy:
- Delivers precise results with high sensitivity.
- Exhibits rapid reaction, enabling clear differentiation between positive and negative samples, particularly in cases of weakly positive specimens.
– Convenience:
- Does not require cold-chain preservation during transportation.
- Minimizes the risk of leakage or spillage during transit.
Specification
| Sample types | Gastric biopsy sample or culture medium |
| Sensitivity | 100% |
| Specificity | 100% |
| Accuracy | 100% |
| Time | 30 minutes |
| Level of asepsis | No presence of other microorganisms |
| Storage | 2 – 8oC, 06 months from the date of manufacture |
| Kit components | Urease test, Sterile water, Sample transfer swab, Sample identification label |
Result analysis
| Reagent color | Results | Note |
| Changes from yellow to deep pink within 5–10 minutes.
| Positive | The sample containing H. pylori exhibits high enzymatic activity. |
| Changes from yellow to red or reddish‑pink within 30 minutes.
| Samples with a low level of H. pylori or patients with regressed H. pylori infection after prior treatment. | |
| Changes from yellow to yellow‑orange after 30 minutes.
| Negative | Color change may result from samples containing very low levels of H. pylori, contamination with Proteus or Morganella species, or temporary effects caused by gastric pH. |
| Medium remains yellow after 30 minutes..
| Negative |

Figure 1. Reagent sample after performing the reaction.
1: Before reconstitution
2: After reconstitution
3: Reagent sample with a negative reaction
4: Reagent sample with a positive reaction
5: Reagent sample with a weak positive reaction
Read more: click here
Read more: Instructional Video for Qualitative Real-time PCR Kit




Reviews
There are no reviews yet.